Every cell in your body has a maintenance crew. Not a metaphorical one, but a real biological process that identifies damaged components, breaks them down, and recycles the parts into raw materials your cells can use to repair and rebuild themselves. This process is called autophagy (from the Greek words meaning "self-eating"), and it won a Nobel Prize in 2016 for good reason.
Autophagy is a fundamental survival mechanism your body relies on every day. When it's working properly, your cells stay clean and efficient. Damaged proteins get cleared. Dysfunctional mitochondria get recycled. Cellular debris that would otherwise accumulate and impair function gets broken down and repurposed. It's essentially your body performing its own deep clean at the cellular level, around the clock, without you ever thinking about it.
When autophagy is impaired, that waste can accumulate. Cells become less efficient, more prone to dysfunction, and more vulnerable to disease. Research has linked impaired autophagy to neurodegenerative conditions, cancer, cardiovascular disease, metabolic dysfunction, and accelerated biological aging.
But here's what makes autophagy particularly relevant for anyone focused on their longevity and healthspan. Unlike many cellular processes, autophagy responds directly to how you live. When you eat, how you exercise, and how well you sleep directly influence whether this system is running efficiently or being suppressed.
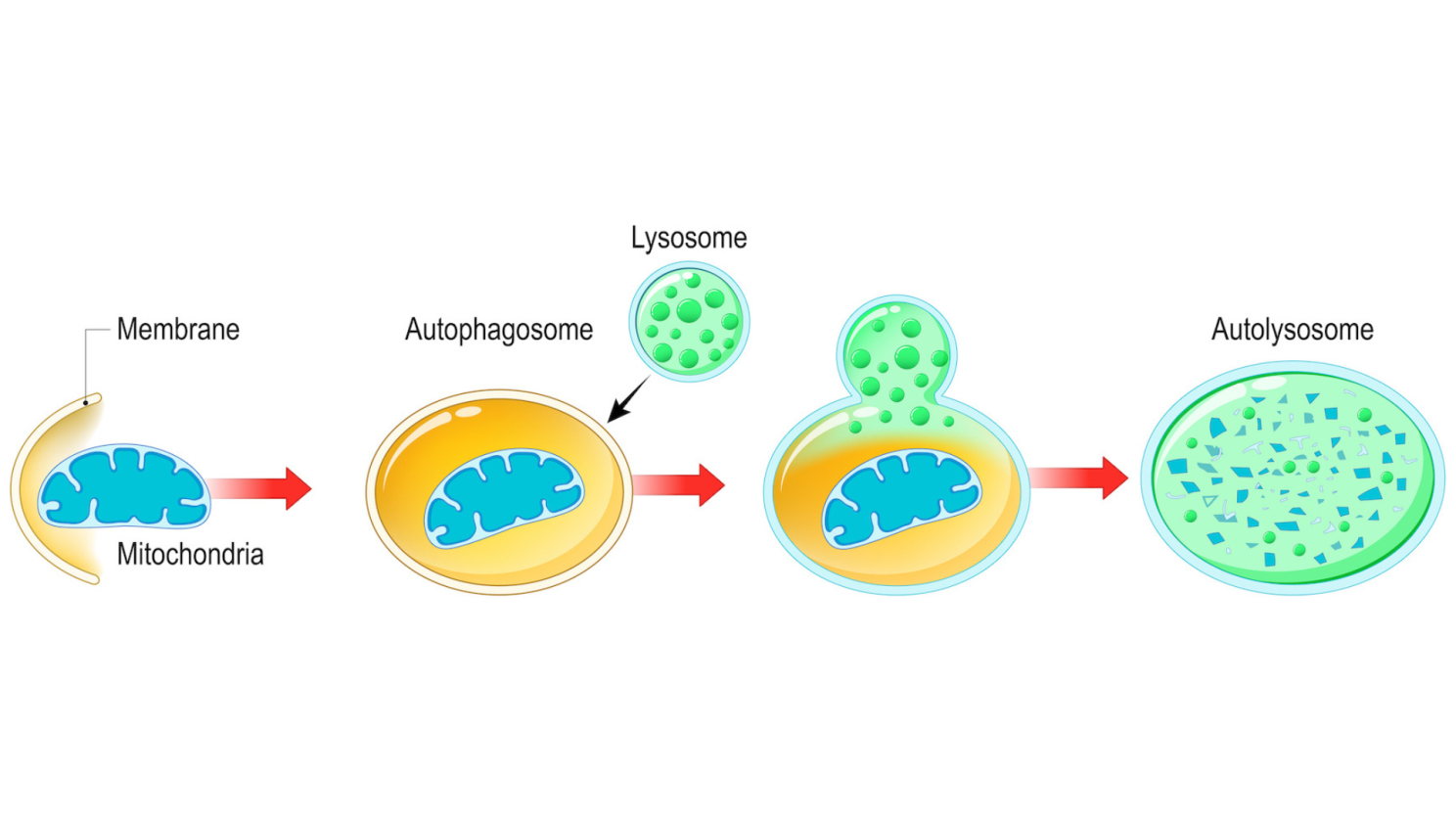
Autophagy begins when your cells detect damaged or dysfunctional components: misfolded proteins, damaged mitochondria, broken organelles (the specialized structures inside your cells that perform specific functions, similar to tiny organs within each cell), and accumulated debris such as aggregated proteins and cellular waste products that build up from normal metabolic activity. Once identified, a double-membrane structure called a phagophore forms around the targeted material. Think of the phagophore as a biological trash bag that wraps itself around the damaged parts, where it then gradually expands until it completely engulfs the damaged components. This creates a sealed compartment called an autophagosome which is essentially a package containing everything marked for recycling.
The autophagosome then fuses with a lysosome which can simply be described as an organelle that functions as your cell's digestive system. Lysosomes contain enzymes that break down the contents into basic building blocks: amino acids, fatty acids, and nucleotides. These raw materials are released back into the cell where they can be used to build new proteins, generate energy, and construct new cellular structures.
Whether autophagy is active or suppressed in your cells comes down to two molecular sensors that act like a switch.
Both states are necessary. You need mTOR for muscle building, tissue repair, and growth. You need AMPK and autophagy for cellular cleanup and maintenance. The problem arises when constant eating, inactivity, and excess caloric intake keep mTOR chronically activated, leaving your cells stuck in growth mode with little opportunity for the cleanup they need.
Brain Health: Neurons are among the longest-lived cells in your body and largely cannot be replaced. This makes efficient waste removal critical. Neurodegenerative diseases like Alzheimer's and Parkinson's are characterized by the accumulation of misfolded proteins that healthy autophagy would normally clear. Research has shown that autophagy dysfunction tends to be present in the early stages of multiple neurodegenerative conditions.
Cancer Prevention: In healthy cells, autophagy can function as a tumor suppressor by clearing damaged DNA, dysfunctional organelles, and reactive oxygen species that may lead to cancerous mutations. Research has found that when autophagy genes are impaired, cells tend to accumulate more DNA damage and may be more susceptible to malignant transformation. It's worth noting that this relationship is complex, as some established tumors may also use autophagy to survive under stressful conditions.
Metabolic Health: Autophagy can help maintain insulin sensitivity by clearing dysfunctional mitochondria and reducing intracellular lipid accumulation (the buildup of fat droplets inside cells that interferes with their normal function) in the liver and muscle. When autophagy is impaired, metabolic waste can build up in these tissues, contributing to insulin resistance, fatty liver disease, and metabolic dysfunction.
Immune Function: Autophagy can help clear intracellular pathogens (bacteria and viruses that invade and live inside your cells), regulate inflammatory responses, and support immune cell function. When impaired, inflammatory responses can become chronic rather than resolving appropriately. This connection may be one reason why chronic inflammation tends to increase with age as autophagy naturally declines.
Cellular Aging: Autophagy can directly influence how quickly your cells age by clearing damaged mitochondria (a process called mitophagy) and helping remove senescent cells (the damaged cells that stop dividing but remain metabolically active, releasing inflammatory signals that can harm surrounding healthy tissue). Research on long-lived populations has consistently found that more efficient autophagy is associated with longer and healthier lifespans.
Fasting: You've probably heard that fasting is good for you. But has anyone explained why at the cellular level? When you stop eating for an extended period, your body doesn't just burn stored fat for energy. When you stop eating, insulin levels drop, mTOR activity decreases, and AMPK activates, shifting your cells into cleanup mode. Research suggests autophagy tends to increase significantly after approximately 14 to 16 hours of fasting, with more substantial activation around 24 to 48 hours. This timeline overlaps with the point at which your body increasingly shifts toward ketosis (when your liver begins converting fatty acids into ketone bodies for fuel because glucose is no longer readily available). This metabolic shift and the rise in ketone levels appear to further stimulate autophagy, which is one reason why the combination of fasting and ketosis has gained so much attention in longevity science. You don't need extended multi-day fasts to benefit. Even condensing daily food intake into an 8 to 10 hour window can provide regular autophagy activation as part of your routine.

Exercise: Physical activity depletes cellular energy (activating AMPK), can create mechanical stress, and can generate mild oxidative stress, all of which can trigger autophagy. Both aerobic and resistance training appear to activate it, though higher-intensity exercise seems to stimulate autophagy more.
Sleep: During sleep, your body enters a natural fasting state that promotes autophagy. Sleep also triggers increased activity of the glymphatic system (your brain's waste clearance system) that works alongside autophagy to help clear toxic proteins from neural tissue. This is one reason why consistently poor sleep doesn't just leave you tired. It can rob your cells of some of their most important maintenance hours.
Specific Compounds: Coffee (both caffeinated and decaffeinated) contains polyphenols that may help activate autophagy. Green tea contains a compound called catechin EGCG (epigallocatechin gallate) that has demonstrated autophagy-promoting effects in research. Resveratrol (found in grapes and berries) and spermidine (found in aged cheese, mushrooms, whole grains, and legumes) have also shown autophagy-enhancing properties in studies.
Constant Eating and Snacking: Every time you eat, insulin rises (the hormone that signals your cells to absorb glucose from your bloodstream) and mTOR activates. Eating every two to three hours from morning to night keeps mTOR chronically active and autophagy never gets a meaningful window to operate. For many people, this alone may be the biggest reason their autophagy is impaired, and they don't even realize it.
Sedentary Behavior: Physical inactivity fails to generate the energy depletion and cellular stress signals that can trigger autophagy. Without regular movement, AMPK remains underactivated, mTOR stays dominant, and your cells miss one of the most consistent daily opportunities for cellular cleanup.
Chronic Sleep Deprivation: Poor sleep disrupts the natural fasting state that sleep provides and can impair the brain's glymphatic waste clearance system. Sleep deprivation also tends to elevate cortisol and insulin levels, both of which suppress autophagy. Over time, this means your brain and body may lose critical hours of cellular maintenance every night.
Chronic Stress: Sustained psychological stress and chronically elevated cortisol can dysregulate autophagy and contribute to cellular damage. While acute, short-term stress can actually activate protective autophagy responses (this is part of why exercise works), chronic stress creates persistent hormonal imbalance that can impair the normal autophagy cycle.
Autophagy naturally becomes less efficient as you age. The molecular machinery slows down, meaning damaged components can accumulate faster and get cleared more slowly. This decline is considered one of the hallmarks of biological aging, and it may help explain why the risk of neurodegenerative disease, cancer, and metabolic dysfunction all tend to increase with age.
This is why the lifestyle factors that support autophagy become increasingly important over time. The habits that activate it are the same habits that research consistently associates with healthier aging and longer healthspan. The earlier you build these habits, the longer your cells benefit from them.
Autophagy is your body's built-in quality control system at the cellular level. It can help clear damage, recycle resources, support defense against disease, and directly influence how your body ages. The remarkable part is that you don't need a prescription, a supplement, or expensive testing to support it. Your body already has the machinery. It just needs the right conditions to run.
Create regular fasting windows. Exercise consistently, including higher-intensity work. Prioritize quality sleep. Eat whole, nutrient-dense foods. Minimize constant snacking. These aren't radical interventions. They're the same foundational habits that support virtually every aspect of health, and autophagy is one of the key cellular mechanisms explaining why they work.
Your cells are designed to maintain themselves. Give them the opportunity, and they will.
