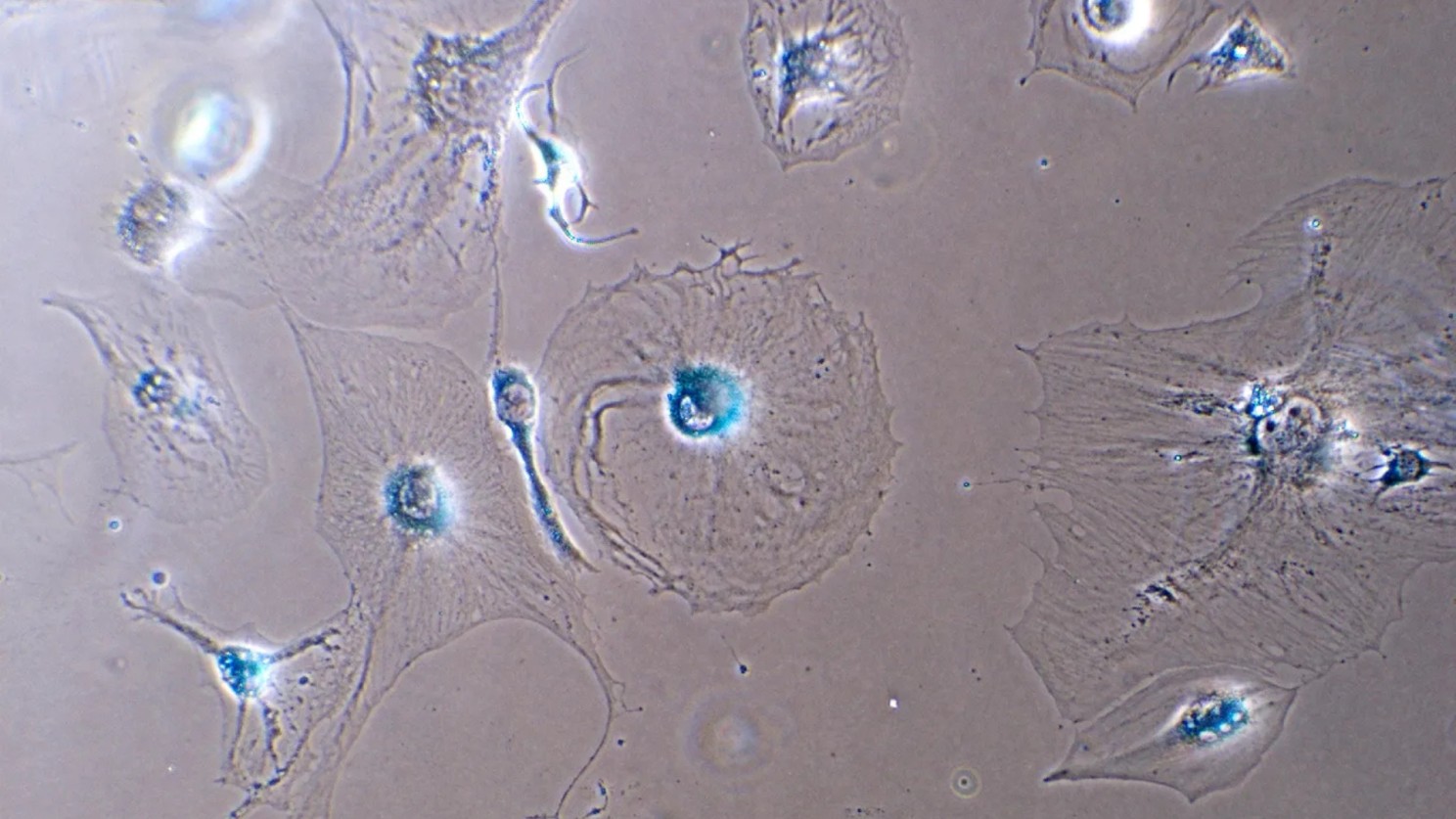
Not all cells in your body are doing their job. Some have stopped dividing permanently. They can't regenerate tissue and they don't respond to normal signals. They've entered a state called cellular senescence, and instead of quietly fading away, they actively damage the tissues around them.
Unlike healthy cells that divide and function normally, or damaged cells that trigger their own death through apoptosis (programmed cell death), senescent cells occupy a middle ground. They're alive but dysfunctional, resistant to the signals that would normally clear them, and metabolically active in ways that harm surrounding tissue.
Senescent cells are both necessary and destructive. During development and wound healing, temporary senescence helps orchestrate tissue remodeling. But when senescent cells persist and accumulate, they become a key driver of many age-related diseases.
Understanding senescent cells reveals why aging isn't just about time passing. It's about cellular damage accumulating faster than your body can clear it.
Cellular senescence is a state of permanent cell-cycle arrest that occurs in response to various stressors including DNA damage, oxidative stress (damage caused by unstable molecules that harm cells), telomere shortening (the gradual wearing down of protective caps on chromosomes), oncogene activation (when genes that can cause cancer become turned on), and chronic inflammation. When cells become senescent, they stop dividing but remain metabolically active, meaning they continue consuming energy and producing molecules even though they're no longer functioning properly.
The defining feature of senescent cells isn't just their inability to divide. It's what they do instead. Rather than quietly existing in a dormant state, senescent cells actively secrete a complex mixture of inflammatory cytokines (signaling molecules that trigger inflammation), proteases (enzymes that break down proteins and tissue), and growth factors (molecules that stimulate cell growth). This secretory profile is known as the senescence-associated secretory phenotype (SASP), and it's the primary mechanism through which senescent cells damage surrounding tissue.
The SASP includes pro-inflammatory molecules like interleukins (immune signaling proteins that promote inflammation), tumor necrosis factor (a protein that drives inflammatory responses), and matrix metalloproteinases (enzymes that break down the structural framework holding tissues together). This inflammatory output doesn't stay localized. It can create a toxic microenvironment that induces senescence in neighboring cells, which creates a self-perpetuating cycle of cellular dysfunction.
Senescent cells also develop resistance to apoptosis (programmed cell death), which can be described as the normal process by which damaged or unnecessary cells are eliminated. This resistance allows them to persist in tissues despite their dysfunctional state, accumulating over time as the immune system's ability to clear them declines with age.
In youth, senescent cells are typically cleared efficiently by the immune system, particularly by natural killer (NK) cells and macrophages (specialized immune cells that engulf and destroy damaged or dysfunctional cells). This clearance maintains tissue health and prevents excessive accumulation.
However, several factors can contribute to senescent cell accumulation as we age:
By age 60, tissues can contain substantial numbers of senescent cells. They accumulate in adipose tissue (body fat), blood vessels, joints, skeletal muscle, the liver, kidneys, and even the brain. Their presence correlates with nearly every age-related disease including cardiovascular disease, type 2 diabetes, osteoarthritis, neurodegenerative diseases, and cancer.
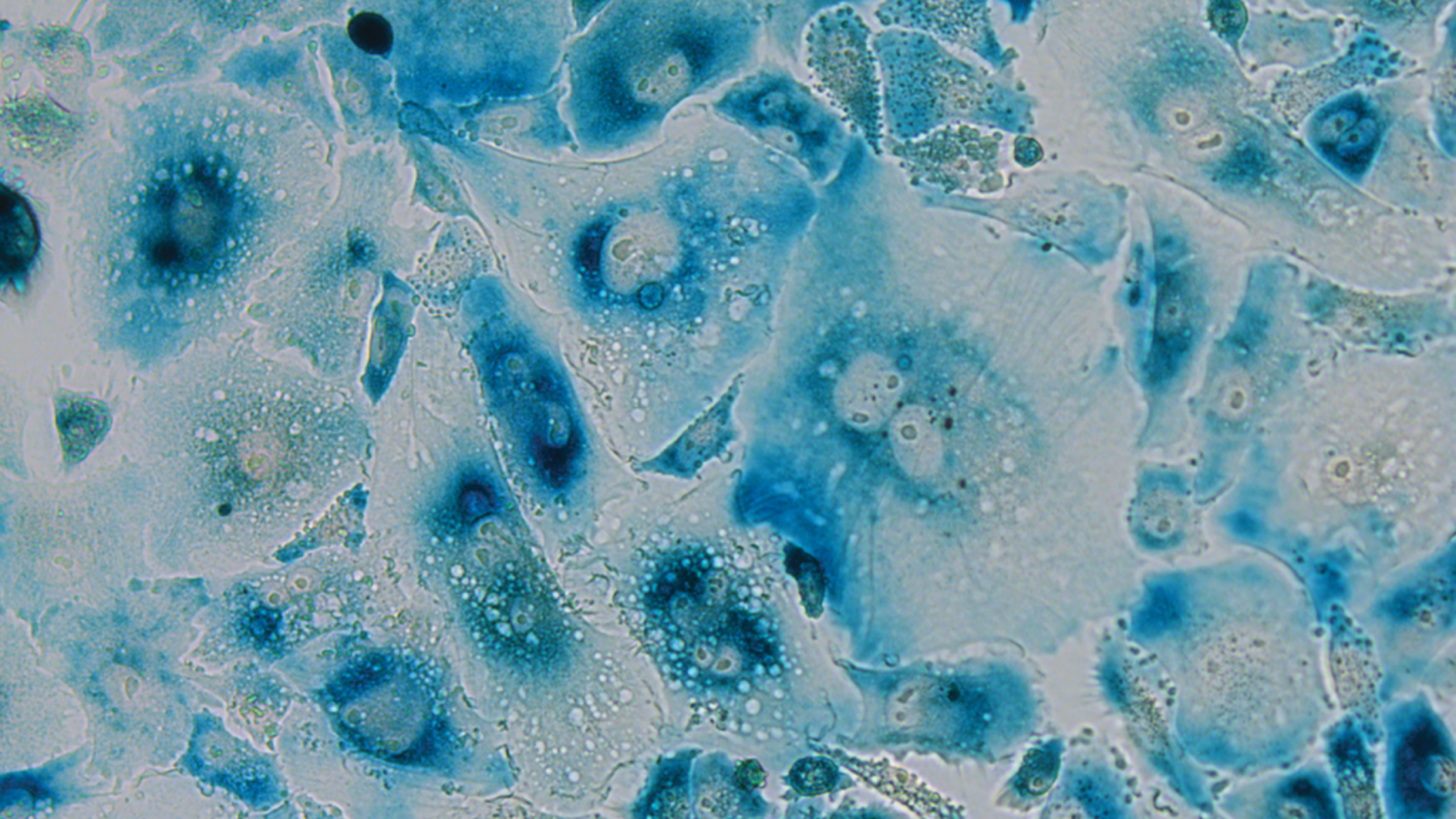
It's important to recognize that cellular senescence isn't purely harmful. Temporary, controlled senescence serves essential biological functions.
During embryonic development (the stage when an organism grows from conception to birth), senescence plays a role in tissue patterning and organ formation. In wound healing, senescent cells can help orchestrate tissue repair by secreting factors that recruit immune cells and promote tissue remodeling. Senescence can also act as a tumor suppression mechanism by preventing damaged cells with oncogenic mutations (genetic changes that can lead to cancer) from dividing uncontrollably.
The problem isn't senescence itself. It's the chronic persistence and excessive accumulation of senescent cells beyond what the body can manage. When senescent cells linger in tissues for extended periods, their beneficial signaling becomes chronic inflammation and their protective role becomes pathological, meaning it shifts from helpful to harmful.
The SASP creates chronic low-grade inflammation throughout the body, contributing to cardiovascular disease by promoting atherosclerosis, vascular stiffness, and endothelial dysfunction (damage to the inner lining of blood vessels that impairs their ability to regulate blood flow and pressure). Senescent cells accumulate in adipose tissue where they can contribute to insulin resistance and metabolic dysfunction. In joints, senescent chondrocytes (cartilage cells) drive osteoarthritis by degrading the structural framework of cartilage and promoting inflammation.
In the brain, senescent glial cells may contribute to neuroinflammation and neurodegenerative diseases. Senescent cells in the kidneys can impair filtration and contribute to chronic kidney disease. In the lungs, they're implicated in idiopathic pulmonary fibrosis (progressive scarring of lung tissue that makes breathing increasingly difficult) and chronic obstructive pulmonary disease (COPD).
Perhaps most significantly, senescent cells tend to create an environment that promotes cancer development even though senescence initially evolved as a tumor suppression mechanism. The chronic inflammation and growth factors in the SASP can paradoxically support tumor growth, angiogenesis (the formation of new blood vessels that feed tumors), and metastasis (the spread of cancer to other parts of the body) in some contexts.
While senescent cells accumulate naturally with age, research suggests that several lifestyle interventions and emerging therapies may help reduce their burden and mitigate their harmful effects.
Regular Exercise
Aerobic exercise is one of the most consistently demonstrated methods for reducing senescent cell accumulation. Exercise works through multiple mechanisms: it improves immune function and enhances the body's ability to clear senescent cells, reduces chronic inflammation to create a less favorable environment for senescence, promotes autophagy to help cells clear damaged components, and reduces oxidative stress.
In humans, physically active individuals show lower levels of inflammatory markers associated with the SASP. Research showing benefits typically involves moderate to vigorous aerobic exercise performed regularly, such as at least 150 minutes per week of moderate intensity or 75 minutes of vigorous intensity.

Intermittent Fasting and Caloric Restriction
Time-restricted eating and periodic fasting may help reduce senescent cell burden through several mechanisms. Fasting triggers autophagy, helping remove damaged components and potentially senescent cells. It creates metabolic stress that may selectively disadvantage senescent cells while supporting healthy cell survival. Fasting also reduces insulin and IGF-1 (a growth hormone that declines with age) signaling, pathways implicated in cellular senescence.
Animal studies show that caloric restriction and intermittent fasting reduce senescent cell markers and extend healthspan. Human evidence is still emerging, but studies suggest that intermittent fasting reduces inflammatory markers and improves metabolic health. Approaches like 16:8 time-restricted eating (16 hours fasting, 8-hour eating window) or periodic 24-hour fasts show promise, though more research in humans is needed.
Managing Chronic Inflammation
Since chronic inflammation both promotes senescence and is amplified by the SASP, reducing inflammatory burden may help limit senescent cell accumulation. An anti-inflammatory diet rich in omega-3 fatty acids (from fatty fish), polyphenols (from berries, green tea, dark chocolate), and fiber while limiting processed foods, added sugars, and excessive omega-6 fatty acids can help. Adequate sleep (7 to 9 hours) is critical for managing inflammation. Stress management through meditation, mindfulness, or other techniques reduces cortisol and inflammatory signaling.
Optimizing NAD+ Levels
NAD+ (nicotinamide adenine dinucleotide) declines with age and is involved in cellular energy metabolism, DNA repair, and sirtuins (proteins involved in cellular health and longevity). Some evidence suggests that supporting NAD+ levels may help reduce cellular stress linked to senescence, though definitive human data are still limited. NAD+ precursors like nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) are being studied. Exercise, fasting, and maintaining metabolic health naturally support NAD+ levels.
Emerging Senolytic Therapies
Senolytics are compounds that selectively induce apoptosis in senescent cells, effectively clearing them from tissues. This is an active area of research with several candidates in clinical trials.
For example, dasatinib (a cancer drug) combined with quercetin (a plant flavonoid) has shown promise in early human trials for clearing senescent cells and improving physical function in older adults. Fisetin, a flavonoid found in strawberries and other fruits, shows senolytic properties in animal studies and is being tested in human trials. Other compounds including navitoclax (a targeted cancer therapy) and BCL-2 inhibitors (drugs that help trigger cell death) are being investigated for their senolytic potential.
It's important to note that senolytic therapies are still very experimental. While some compounds like quercetin and fisetin are available as supplements, their effectiveness, optimal dosing, and long-term safety in humans for senolytic purposes remain under investigation. These should not be considered established interventions at this time.
Senomorphics (SASP Inhibitors)
Rather than killing senescent cells, senomorphic compounds aim to suppress the harmful SASP without eliminating the cells themselves. Compounds like rapamycin and related drugs (rapalogs), metformin (a diabetes medication), resveratrol, and EGCG (from green tea) are being investigated for SASP-suppressing effects, primarily supported by preclinical and early clinical studies.
Like senolytics, senomorphics are still largely experimental, and their use outside of clinical trials or established medical indications should be approached cautiously.
Senescent cells don't suddenly appear at age 60. They begin accumulating gradually throughout your life, with the rate of accumulation accelerating as immune clearance declines and cellular damage increases.
In your 20s and 30s, maintaining strong immune function and minimizing cellular damage through exercise, good nutrition, and stress management can help keep senescent cell burden low. In your 40s and 50s, proactive interventions become increasingly important as natural clearance mechanisms begin to decline and accumulation tends to accelerate. For older adults, reducing senescent cell burden may help slow functional decline and reduce risk of age-related diseases.
The interventions that reduce senescent cell accumulation (regular exercise, intermittent fasting, anti-inflammatory nutrition, stress management) are largely the same interventions that support overall healthspan (the number of years you live in good health). They work synergistically across multiple aging mechanisms, not just senescence. Understanding senescent cells and their role in aging provides insight into how cellular health influences the aging process. The research on senescent cells continues to evolve, revealing that many aspects of cellular aging respond to the choices we make every single day!